Are bigger hospitals safer than smaller hospitals when it comes to surgical instrument reprocessing? Not necessarily, it turns out, because those hospitals often have more complex service offerings that can complicate and burden the reprocessing workload.
“Far too often, bigger hospitals mean bigger potential for the critical needs of sterile processing teams to go unmet,” says Hank Balch, founder and president of Beyond Clean, in a recent episode of his podcast.
Balch said he created the episode in response to buzz around a New England Journal of Medicine (NEJM) article reporting that bigger hospitals do not necessarily result in better patient outcomes. It’s a conversation that even stretches across the ocean to England where Louella Vaughan, a senior research fellow, authored “Why Bigger Isn’t Always Better: Caring For Patients in Smaller, Rural and Remote Hospitals,” which appeared in Future Healthcare Journal last year.
Sterile processing departments have long recognized the challenges of bigger organizations, Balch noted in the podcast. Among the problems that can occur when hospitals get bigger:
An opinion piece in General Surgery News notes that surgical site infections are the third most expensive type of healthcare-acquired infection (HAI), costing $21,000 per patient case. It is estimated that the total cost of such infections to the U.S. healthcare system ranges from $3.5 billion to $19 billion annually.
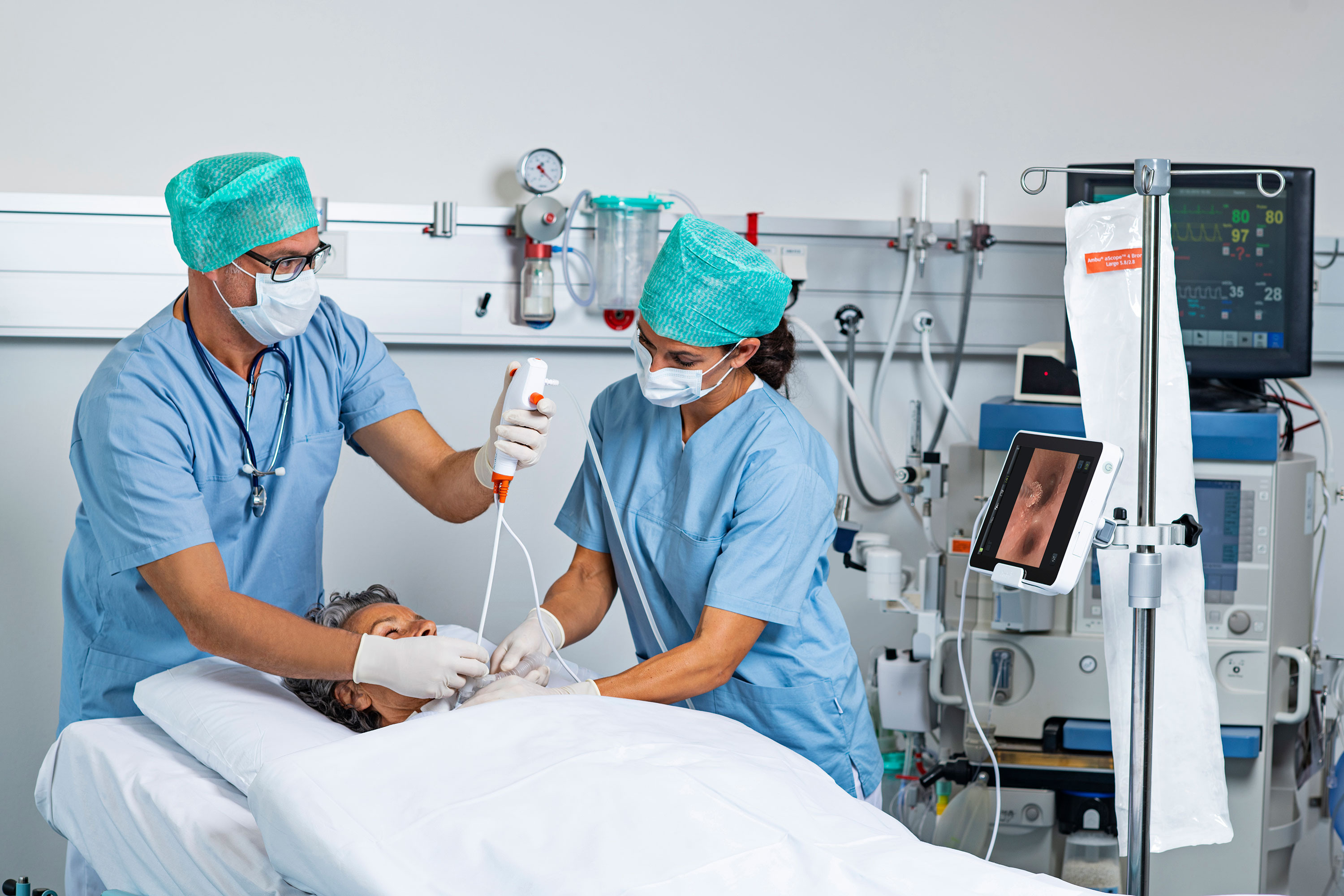
Central sterile processing is the first link in the chain of infection prevention, with the vital role of decontaminating, cleaning, inspecting and sterilizing instruments. Failures in any of those steps can introduce pathogens into the OR and increase risk of infections.
The U.S. Food And Drug Administration recently updated its safety communication and guidance on reprocessing flexible bronchoscopes after medical device reports found nearly an eight-fold increase in bronchoscope-related infections.
The agency has recommended that:
In April, the FDA announced new investigations into numerous MDRs related to urological endoscopes, including some that involved patient infections and three device-related patient deaths outside of the U.S.
Challenges in device reprocessing are related to “the lack of connection that hospital administrators and the public at large make between the resources given to our teams and the ultimate quality of surgical instruments available for every procedure that books into our operating rooms,” Balch says.