An Ohio hospital system says it’s the first in the U.S. to switch to a new, single-use duodenoscope for endoscopic retrograde cholangiopancreatography (ERCP) procedures.
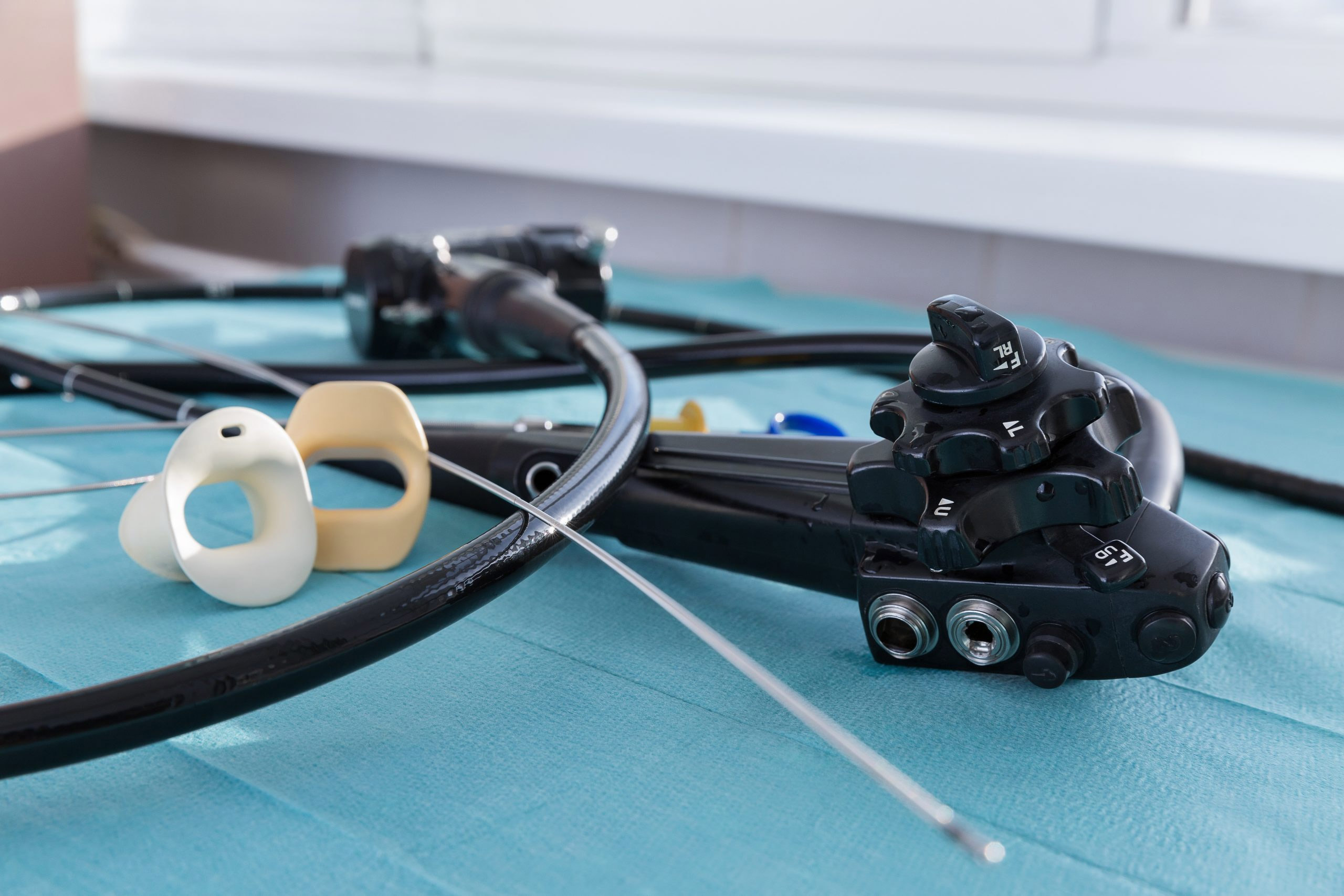
Memorial Health System, based in Marietta, Ohio, is using the aScope Duodeno, developed by Denmark-based Ambu A/S, for ERCP procedures, according to the Marietta Times. This medical device is used to diagnose and treat gallstones, pancreatitis, tumors, liver conditions, and cancer in the bile ducts or pancreas.
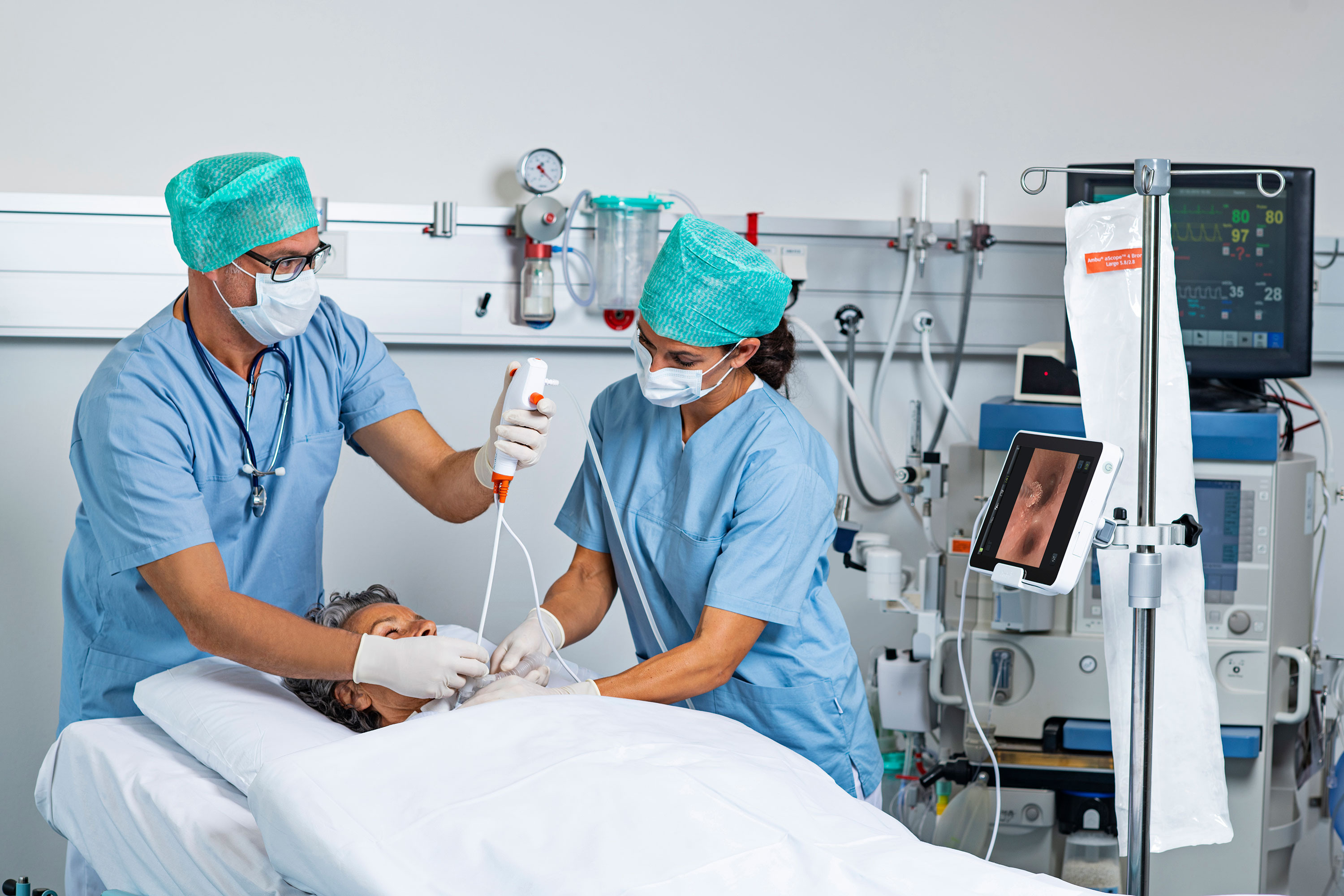
More than 500,000 ERCP procedures — in which the duodenum, the first part of the small intestine beyond the stomach, is examined — are performed annually in the U.S.
Dr. Juan Tejada, a gastroenterologist and advanced endoscopist with Memorial Health, told the Marietta Times the transition to single-use duodenoscopes is “a win for both physicians and patients” by marrying innovative design with a mission to advance patient safety.
Memorial Health is a nonprofit integrated health system that has 500 physicians representing more than 40 specialties and locations across southeastern Ohio and the northwestern part of West Virginia.
Both Ambu and Boston Scientific Corp. have won U.S. Food and Drug Administration approval for fully disposable duodenoscopes in the last 18 months. These complex devices can be difficult to clean; studies show that, while rare, reusable duodenoscopes pose a risk of cross-contamination to patients.
Duodenoscopes have been linked to deadly “superbug” infections, heightening scrutiny not only of the cumbersome reprocessing measures needed to fully clean the scopes but also their design attributes that make cleaning inherently difficult.
In April 2020, the FDA updated its recommendation that duodenoscope manufacturers and healthcare facilities transition to different types of duodenoscopes that may pose less risk to patient safety. That included not just duodenoscopes with disposable components, such as disposable endcaps, but scopes that are fully disposable — an update that came in the wake of Boston Scientific and Ambu receiving regulatory clearance for their single-use devices.
A panel at the 2021 Digestive Disease Week virtual conference explored the question of whether new technology is the best way to address risks of patient cross-contamination in ERCP. Scope performance, cost, and environmental factors will help provide an answer, panelists said.
“Gone are the days when we are doing ERCPs without any thought or regard to the infection risk,” Dr. Mark Gromski of the Indiana University School of Medicine, said during a panel discussion on the adoption of single-use duodenoscopes. “We need a careful, well thought out strategy.”
Gromski spoke with Single-Use Endoscopy in early 2020 about ERCP technology, the cross-contamination issue, and steps healthcare practitioners and manufacturers can take to ensure patient safety.