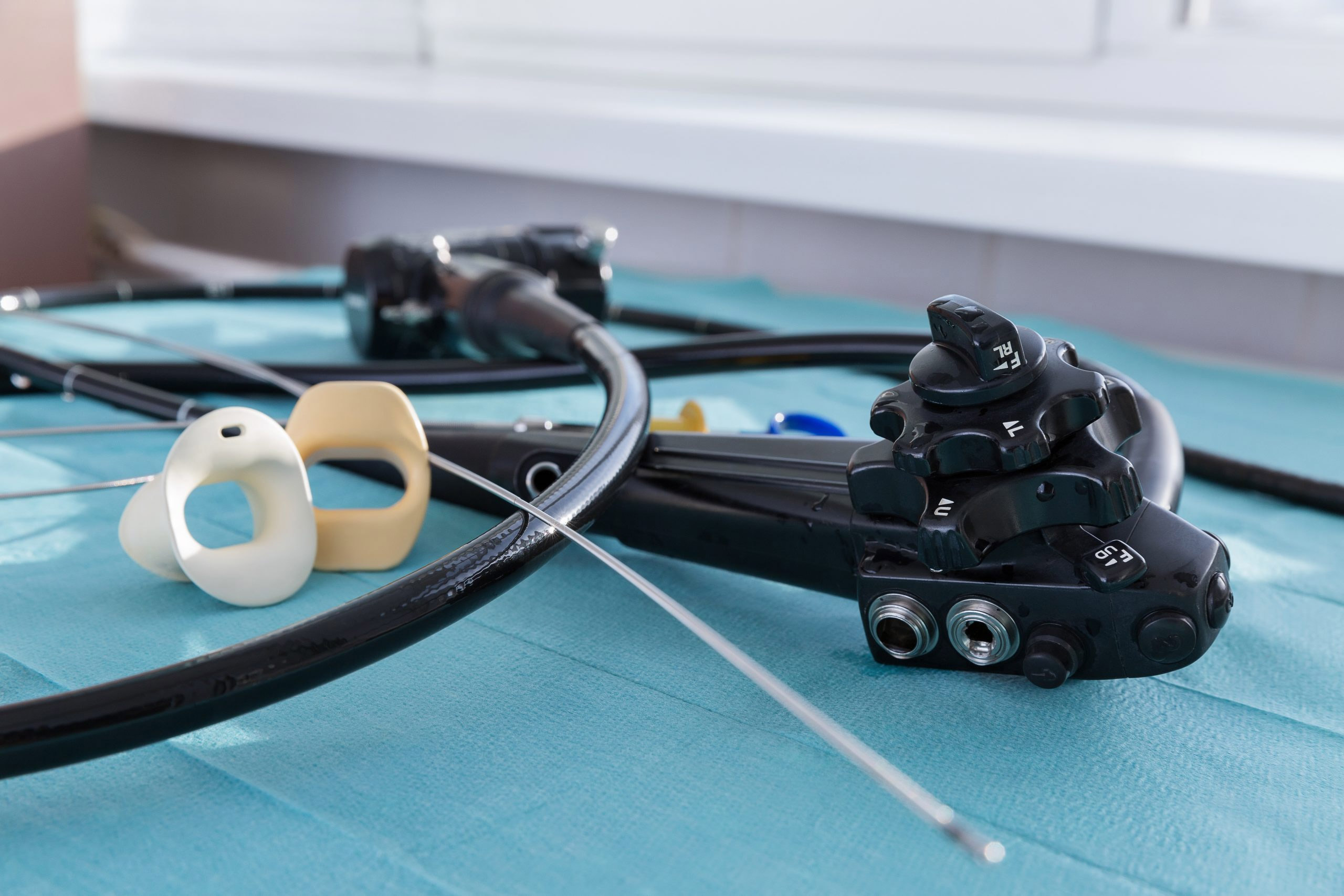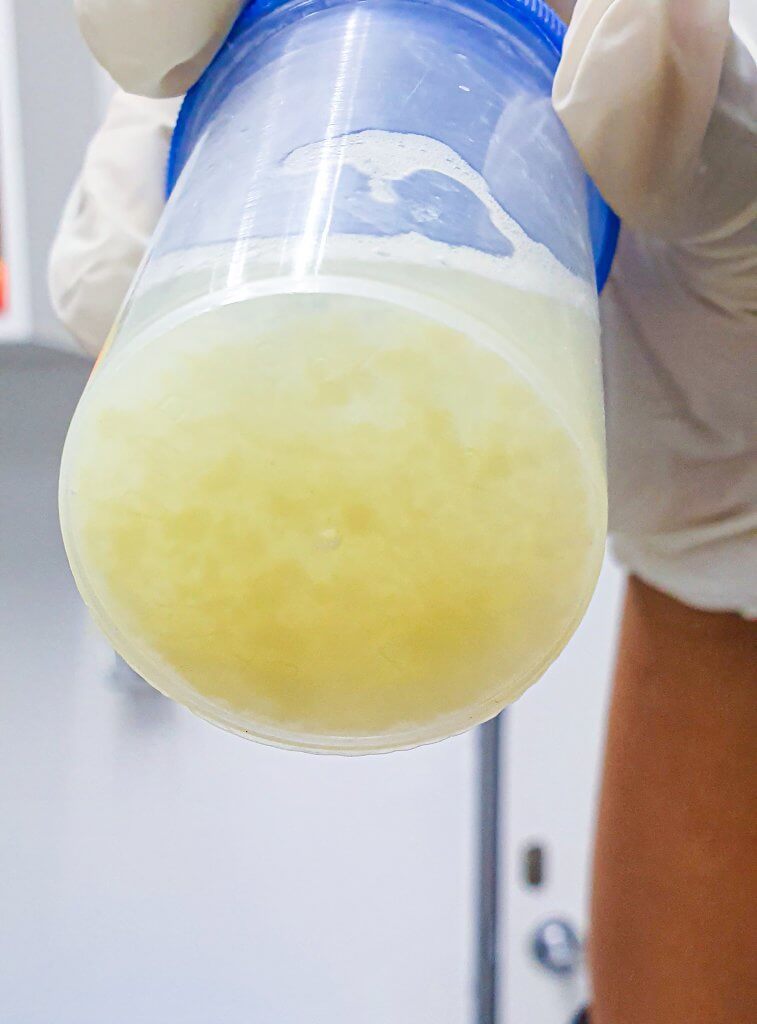
Bronchoalveolar lavage (BAL) and bronchial wash (BW) are very common procedures performed in ICUs around the world to diagnose and treat lung infections.
However, physicians often report losing these fluid samples so vital to diagnosis and treatment due to cumbersome workflows or contaminated tools and accessories, according to a new white paper from Ambu outlining methods to help increase sampling success.
To compile data for this white paper, Ambu surveyed 116 physicians practicing in the ICU and/or bronchoscopy suite in the U.S. and United Kingdom. Losing a BAL sample can cause diagnosis and treatment delays. That's because patients need to recover before the procedure can be performed again.
According to the study, 72 percent of physicians reported having lost a BAL sample. During a bronchoalveolar lavage (BAL), fluid is squirted into a small part of the lung. It's then collected via flexible bronchoscope for examination. The procedure is widely used to diagnose lung disease and ventilator-associated pneumonia (VAP), the second most-common nosocomial infection in the ICU.
Despite the procedure’s common utility for collecting samples in the ICU, BAL remains a cumbersome process, from setup to post-procedure waste disposal.
The paper points to several reasons for the slow and tiresome workflow. That includes the wait time to get a clean flexible endoscope from the reprocessing suite. During the procedure itself, switching between suction and sampling can leave open containers exposed to surrounding elements and contaminants. That can compromise the integrity of the samples collected.
BAL samples may also be spoiled by inadequately reprocessed flexible bronchoscopes that are still harboring infectious organisms. Contaminated bronchoscopes may put a patient at risk of infection and can also jeopardize the integrity of a BAL sample.
In order to ensure BAL success, workflows must be simplified. This will ensure sampling integrity and success.There are approximately half a million bronchoscopies performed in the U.S. each year. The physicians surveyed for this study reported performing up to six BALs every week in the ICU or bronchoscopy suite.
With several physicians in each department, the total number of weekly procedures quickly adds up. This high frequency proves the high clinical value the samples add when diagnosing and treating patients.
With the COVID-19 pandemic heightening concerns of cross-contamination across the U.S., the American Association for Bronchology and Interventional Pulmonology recommended that BAL not be used for lung sampling, unless necessary. Single-use bronchoscopes should be used if bronchoscopy was deemed necessary on a suspected or confirmed COVID-19 patient, the organization said.