.png)
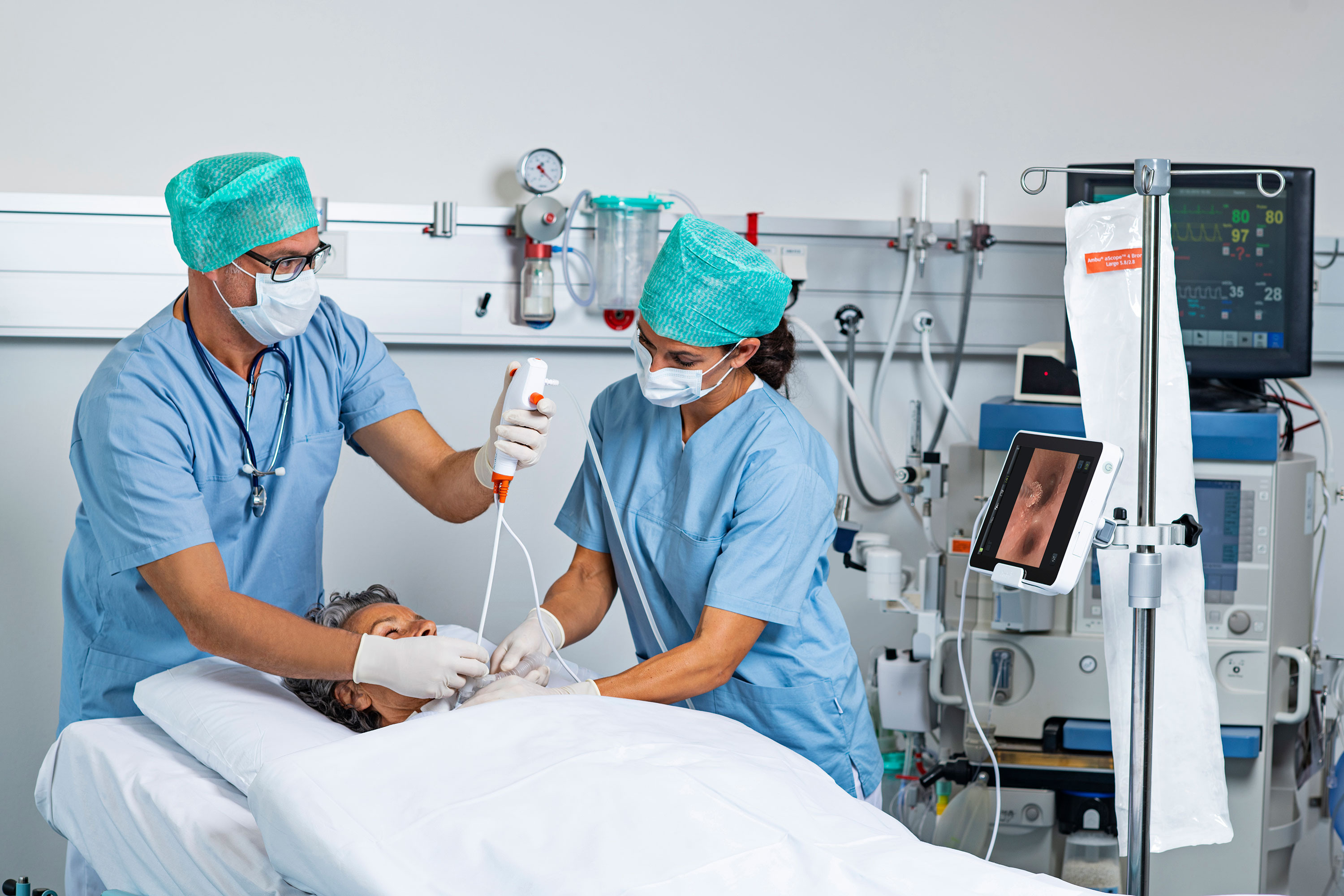
A comprehensive review and analysis of adverse event reports involving flexible endoscopes filed with the U.S. Food and Drug Administration shows a marked increase for six types of scopes from 2014 through 2021.
The analysis was undertaken by Dr. Larry Muscarella, president of LFM Healthcare Solutions and an independent safety expert who advises hospitals, manufacturers and the public about medical device safety and the causes of infections in healthcare facilities. Muscarella combed through the FDA’s so-called MAUDE database — the acronym stands for “Manufacturer and User Facility Device Experience” — and identified reports describing endoscope contamination.
In this conversation, we discuss why this kind of analysis is significant and what the findings reveal about the safe and effective cleaning of endoscopes.
Dr. Muscarella is an Ambu consultant.
Show Notes:
Medtech Insight - Study: FDA Data Shows Endoscope-Related Adverse Events Continue to Rise
FDA Pushes Switch to ‘Innovative’ Duodenoscope Designs to Enhance Patient Safety
Urological Endoscope Manufacturer Issues ‘Urgent’ Recall
------------------------
Endoscopy Insights is a podcast devoted to conversations with thought leaders in flexible and single-use endoscopy. In each episode we’ll explore the ways different clinical and technological innovations are moving the field of endoscopy forward.
Endoscopy Insights is brought to you by Single-Use Endoscopy, part of the Ambu Learning Center. Ambu has been bringing healthcare solutions of the future to life since 1937.