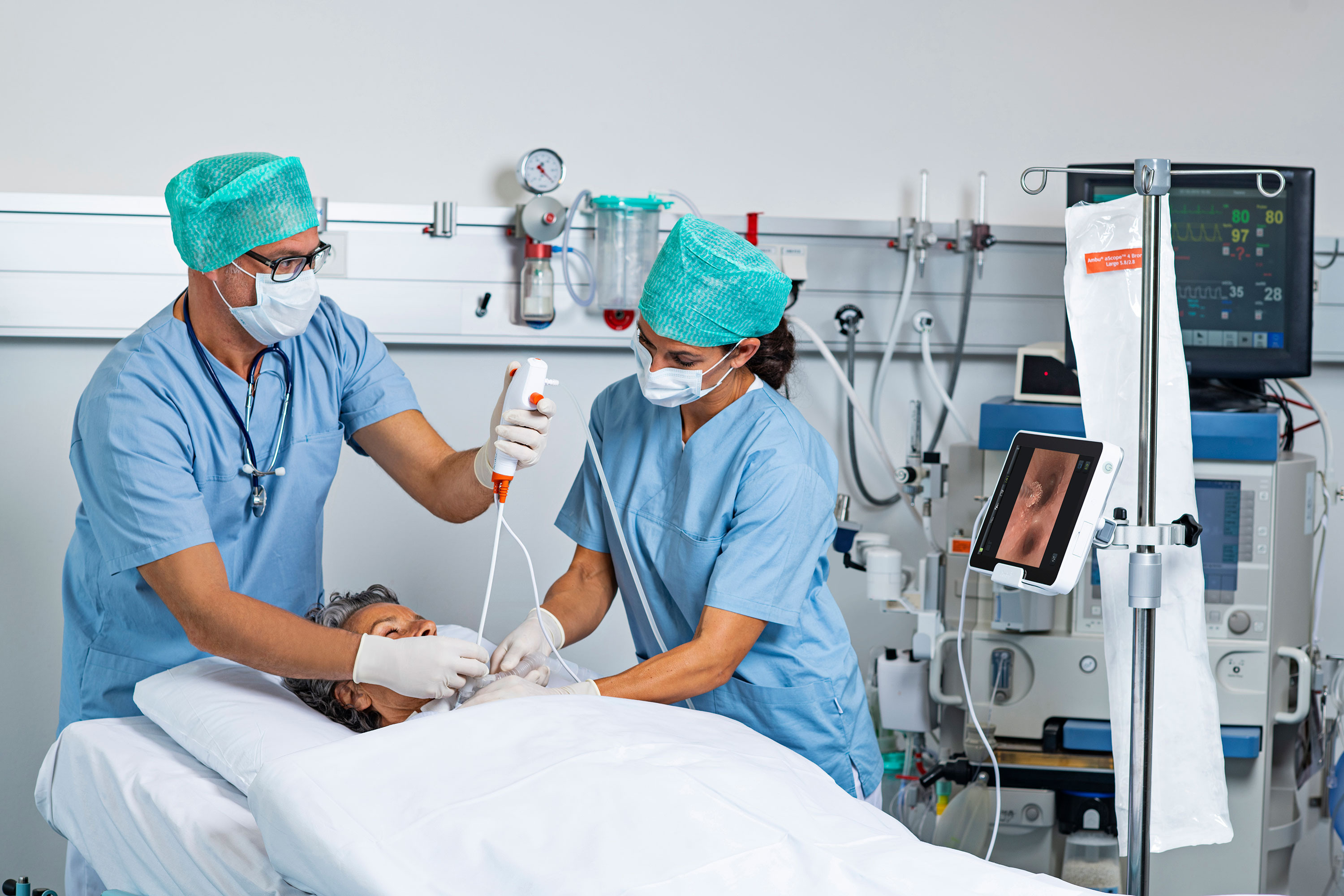
When it comes to disinfecting endoscopes between uses, all patients — regardless of immune system status — should be guaranteed the same level of sterility.
That's according to a panel of experts quoted in a recent review of the January 2021 updates to the multisociety endoscope reprocessing guidelines. The review was published as part of the Gastroenterology & Endoscopy News Priority Report, which is published twice a year.
“Ideally, we want one standard of care; we want it to be consistently high for all patients,” according to Dr. Lawrence Muscarella, an infection prevention specialist and president of LFM Healthcare Solutions. “We don’t really want to start choosing between disinfection or sterilization depending on what we think may be the status of a patient’s immune system, at least not without sound supporting evidence.”
The latest iteration of the multisociety guidelines states that ethylene oxide (EtO) sterilization should be considered for flexible endoscopes in select settings or for specific patient populations. EtO sterilization, which is more expensive than the minimum requirement of high-level disinfection (HLD) for semi-critical devices, is the only method of sterilization available for GI flexible endoscopes.
Dr. V. Raman Muthusamy, the medical director of endoscopy at the University of California, Los Angeles Health and one of the authors of the new guidelines, also weighed in on the sterilization recommendation.
“There are data showing that several institutions that had an outbreak were able to contain and stop the outbreak when they switched to EtO,” Muthusamy told G&E News.
But there is no clinical evidence showing EtO is better than HLD in non-outbreak settings, he added.
The updated multisociety guidance included background and research updates in four key areas of reprocessing: training, intermediate-level disinfection, sterilization vs. high-level disinfection, and the role of alcohol in drying. Also included were recommendations for auditing reprocessing methods and tracking potentially contaminated scopes and the patients on which they were used.
Overall, the recommendations did not change, there was just more explanation provided, according to Dr. Emmanuel S. Coronel of the University of Texas MD Anderson Cancer Center.
There is still, however, a lot to be learned about the most effective endoscope reprocessing methods, he noted. Because of that, the January 2021 version will not be the last, Coronel told G&E News.
For some, like Muscarella, more clarification is still necessary. He argued clearer guidance is needed around which devices can undergo intermediate-level disinfection (not GI flexible endoscopes) and the role of alcohol in the drying step of reprocessing.
Muthusamy said any vagueness around the use of alcohol in drying is due to a general lack of clinical evidence as to the true benefit of using the chemical flush.
“Ultimately, given this uncertainty, we recommend individualizing your response based on what the manufacturer of your device advises,” Muthusamy said. “We would be happy to see further research in this area to make a more definitive recommendation in future guidelines.”