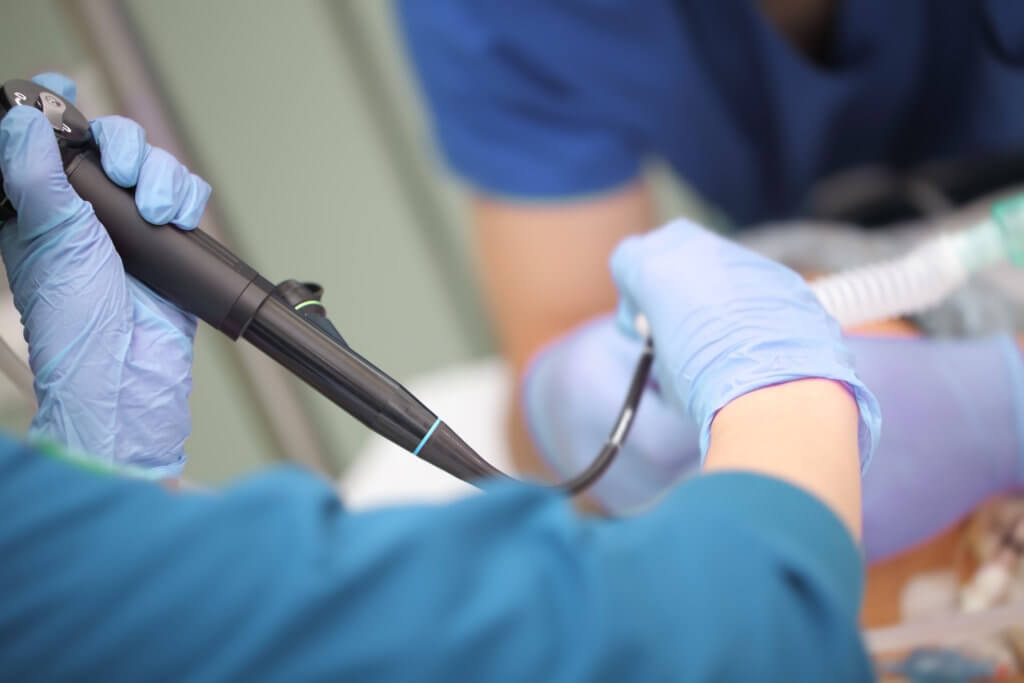
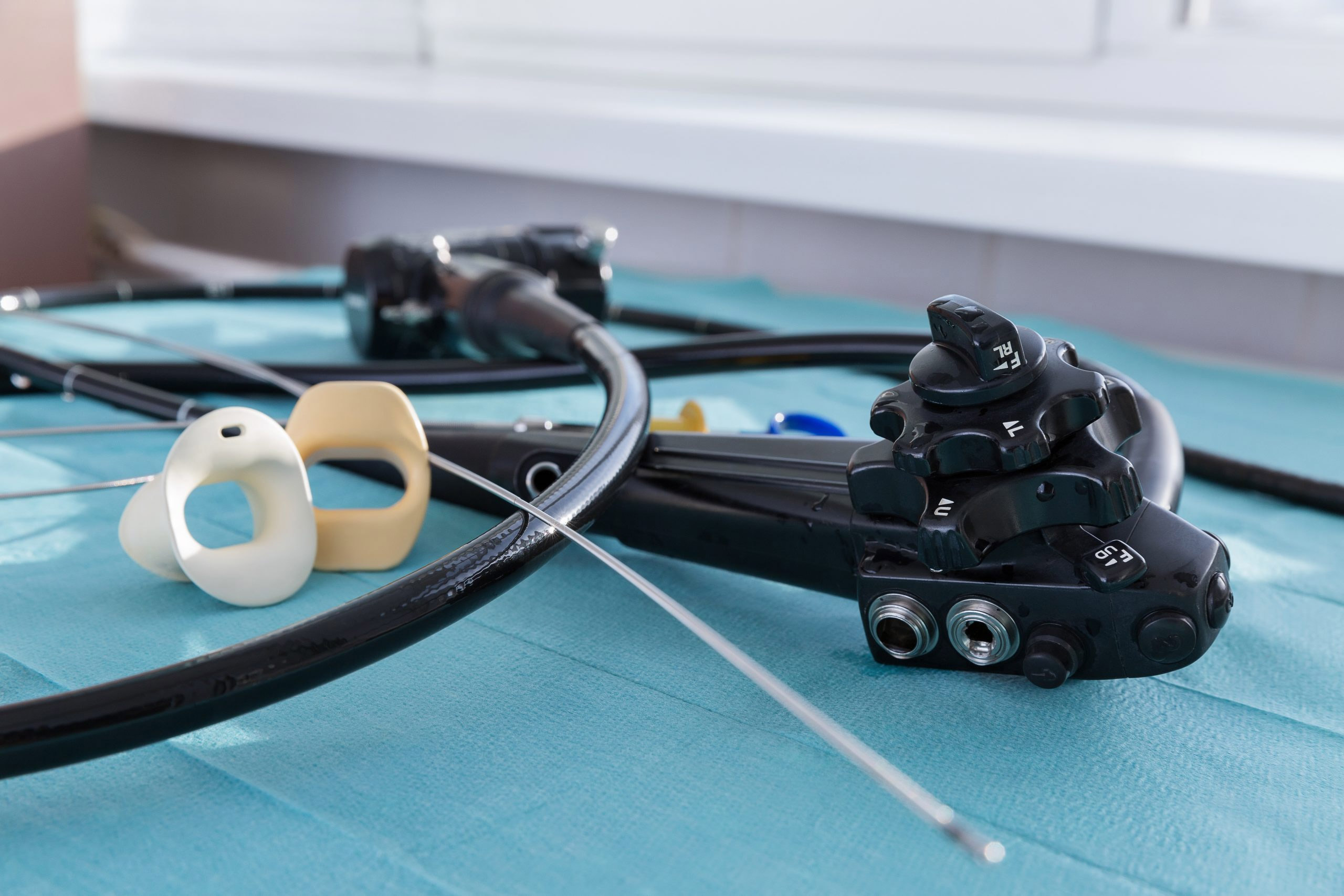
Changing over entire duodenoscope inventories—in the wake of a Food and Drug Administration recommendation that providers who perform endoscopic retrograde cholangiopancreatography (ERCP) move away from fixed endcap duodenoscopes to duodenoscopes with disposable components—will be neither quick, nor straightforward.
For transition plans (if and when they are enacted) to be successful, they must anticipate some of the challenges that may arise in the process. Among the barriers to change each stakeholder may face, costs will be top of mind among providers on two fronts.
First, consider the sunk costs. A single scope can cost about $35,000, depending on the manufacturer. For high-volume facilities, a fleet of duodenoscopes can represent hundreds of thousands in capital expenditures. Any fiscally responsible organization would hesitate to replace such a large inventory.
It remains to be seen, too, how scopes with disposable components would shift the financial burden. Their novelty may translate into a higher per-unit cost. And they will also certainly entail recurring costs, as disposable components are replaced.
Physicians may also be resistant to change—they are, rightly, protective of their workflows. They may push back against any change in their equipment, unless they see a clear benefit to their patients.
The FDA’s surveillance studies, while convincing in the aggregate, are preliminary. Physicians at any particular facility may not accept that such risks apply to their work. Even if they do, they might insist that reprocessing procedures—those conducted by facility sterile processing departments, like manual cleaning or high-level sterilization—should be targeted for intervention, not their tools.
Further, disposable components may entail trade-offs in usability or precision, which any surgeon may be reluctant to embrace. For more on the context and import of the FDA’s recommendations, and for more guidance in making those recommendations a reality at your facility, click here for our overview of the issue and analyses of the impact on manufacturers and infection prevention specialists.