More Urology Articles
How Social Media Benefits Both Urologists and Their Patients
Public Health
Urologists discuss the advantages and disadvantages of social media, which platforms early-career urologists can use to share content and engage with patients, and common pitfalls to avoid.
Triaging Urology Procedures: Proceed With Most Cystoscopies Without Delay
Preventing Infection
Many urological procedures can be safely delayed at this time, but cystoscopy – for diagnostic and surveillance purposes or the removal of ureteral stents – should proceed.
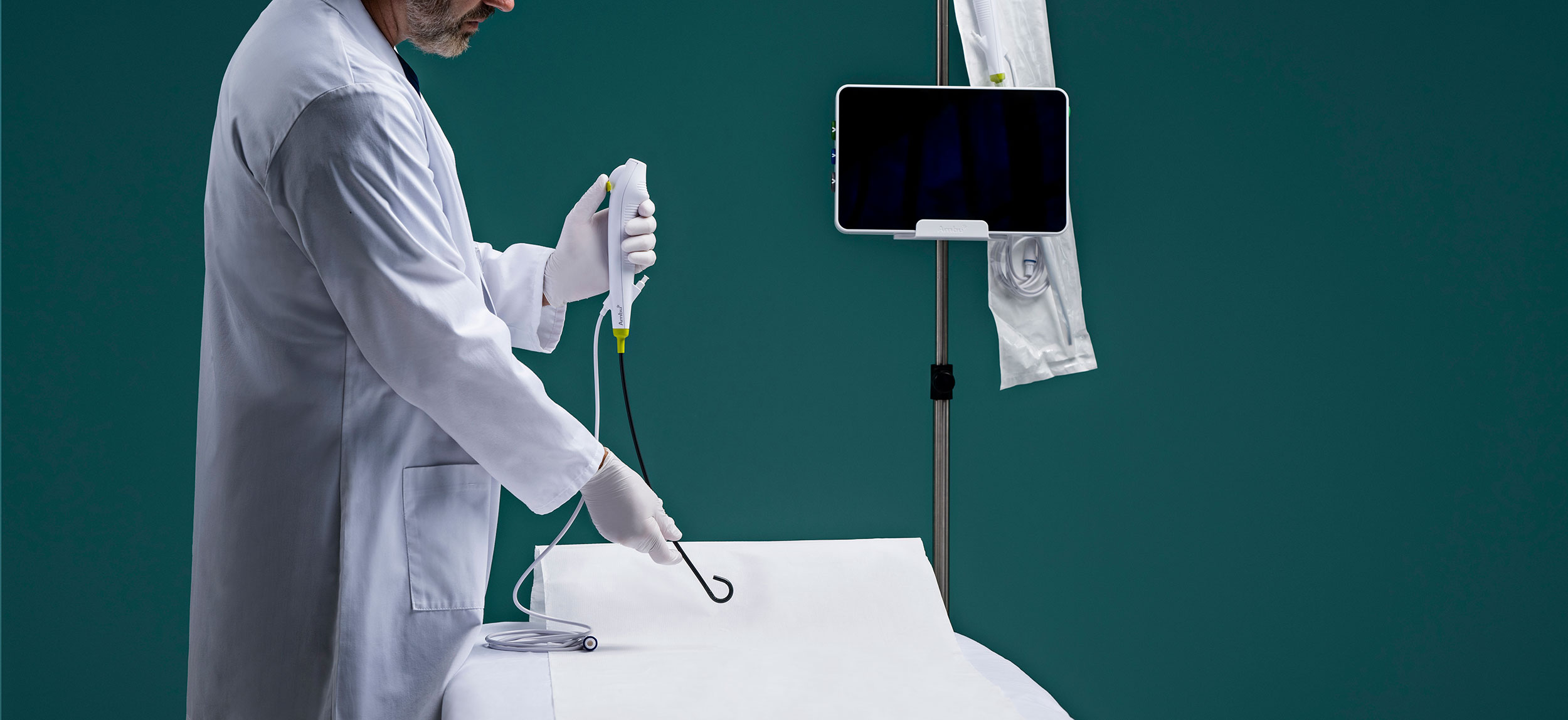
Are Reusable or Single-Use Urological Endoscopes the Choice for Your Facility?
Value-Based Care
The choice between reusable and disposable endoscopes sometimes comes down to economics, according to Outpatient Surgery, an AORN publication.
Infection Outbreak Linked to Reusable Cystoscope Irrigation Plugs
Preventing Infection
Investigators detailed a case at the University Hospitals of Leicester in the UK, where irrigation plugs were the cause of an outbreak pegged to 15 cystoscopy procedures.
AUA 2023: How to Calculate the True Cost of Cystoscopy
Analyzing Value
Research presented at the annual conference detailed how to find the cost per procedure performed with a reusable cystoscope.
More From Single-Use Endoscopy

Environmental Impact
Med-tech and healthcare companies are among those on the prestigious list, recognized for modifying products with sustainability in mind.

Patient Safety
It all starts with your staff and the training they receive.