“Superbugs” are becoming a greater threat to global health as these multidrug-resistant organisms, or MDROs, continue evolving to resist developed antibiotic treatments.
MDROs are also a hot topic in endoscopy as recent media reports of cross-contamination from duodenoscopes have triggered FDA intervention and agency recommendations for disposable components to limit cross-contamination.
But disposable endcaps will not eliminate the total threat of MDROs in endoscopy, even with reprocessing.
In a recent New York Times article about the FDA updates and recommendations, Dr. Erica S. Shenoy, associate chief of infection control at Massachusetts General Hospital, said single-use endcaps, “will reduce, but not entirely eliminate, contamination of devices.” Research has shown that MDROs can thrive in the long channels of endoscopes if protected by biofilm, which acts as a protective barrier against the most commonly-used disinfectants in high-level disinfection.
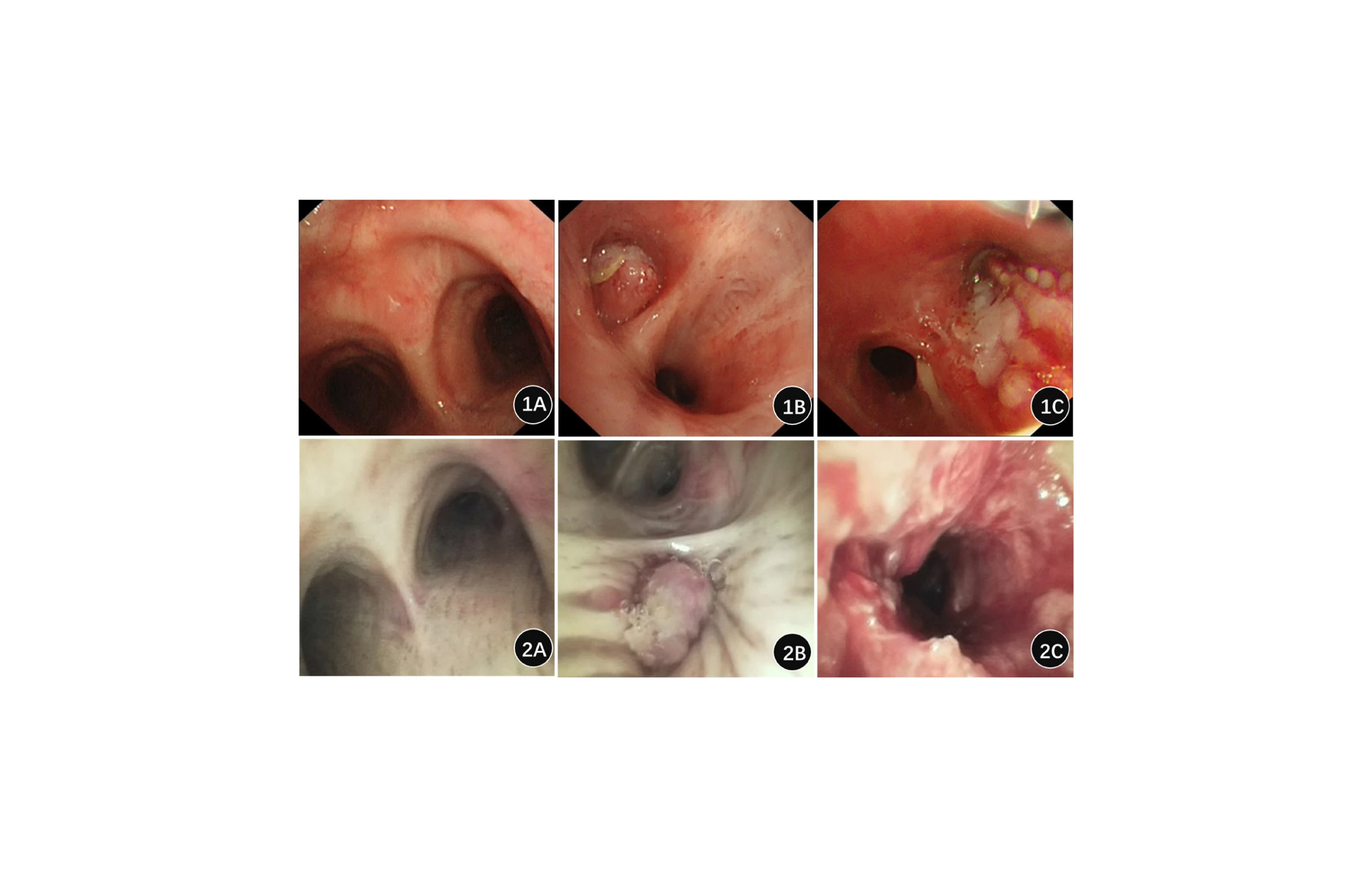
Infectious agents in reported outbreaks from contaminated flexible endoscopes have included bacteria with proven resistance to antibiotics. Pseudomonas aeruginosa, known for its ability to adapt and resist antibiotics even during a course of treatment, is a regular offender in bronchoscope-related outbreaks. P. aeruginosa is one of the most common organisms found in contaminated endoscopes.
In addition, carbapenem-resistant Enterobacteriaceae (CRE), which has been most associated with duodenoscopes and ERCP by the media, has also contributed to a dozen bronchoscope-related outbreaks worldwide since 2012. Dr. Atul Mehta, a pulmonologist at the Cleveland Clinic, and Lawrence Muscarella, president of Pennsylvania-based LFM Healthcare Solutions, conducted a 2019 survey that concluded that CRE and other related MDRO infections in bronchoscopy are more common than currently recognized by the general public.
The bronchoscopes in question, in fact, had been reprocessed according to industry guidelines, according to Mehta and Muscarella’s research. Their findings show a need for stricter reprocessing guidelines and greater awareness about the potential threat of MDRO infection transmission from reprocessed bronchoscopes.
The air/water channels of many endoscopes are too small to be cleaned mechanically. This makes it particularly difficult to prevent the formation of biofilm, as only chemicals can be used. In addition, microscopic cracks, grooves and pits inside these channels can be covered by biofilm.
Biofilm, once formed, is almost impossible to eradicate from the long, narrow internal channels or broken areas of a flexible bronchoscope. The presence of biofilm regularly contributes to reprocessing failures for flexible bronchoscopes because biofilm can harbor and protect bacteria from common disinfectants used during reprocessing.
In the 2013 study, “Transmission of Infection by Flexible Gastrointestinal Endoscopy and Bronchoscopy,” authors Julia Kovaleva, Frans T.M. Peters, Henny C. van der Mei, and John E. Degener concluded that biofilm “allows microorganisms to survive under conditions of drying and chemical and antibiotic exposure.”
In other words, antimicrobial-resistant organisms can survive endoscope reprocessing procedures if they are safely protected by biofilm. “[Biofilm] can create a vicious circle of growth, disinfection, partial killing or inhibition, and regrowth, resulting in outbreaks of endoscopy-related infections in patients who underwent endoscopy with a biofilm-containing endoscope,” according to Kovaleva, and her co-authors.
In a 2017 study, “Tolerance of Pseudomonas aeruginosa in in-vitro biofilms to high-level peracetic acid disinfection,” researchers found peracetic acid was ineffective against bacteria in biofilm.
The authors of the study, A. B. Akinbobola, L. Serry, W.G. Mckay, G. Ramage and C. Williams, concluded that the concentration of disinfectants needed to eradicate the P. aeruginosa biofilm in the experiment was dependent on the age of the biofilm.
According to the study, 96-hour biofilm can adequately protect resistant organisms from the normal amount of disinfectant used to clean bronchoscope lumens in an automated endoscope reprocessor (AER).
Reusable bronchoscopes must be reprocessed via a series of steps requiring both manual cleaning and either high-level disinfection or low-temperature sterilization. Most reusable bronchoscopes cannot tolerate the same disinfectants that other instruments can, so the choices are limited. The FDA recommends bronchoscopes at least undergo high-level disinfection with a liquid disinfectant containing glutaraldehyde, peracetic acid, hydrogen peroxide, and/or ortho-phthaldehyde.
“Unlike antibiotics, which often target specific bacterial pathways, disinfectants have non-specific activity and target multiple cellular components or metabolic pathways simultaneously,” according to Akinbobola and his fellow researchers. “This has led to a relaxed attitude to potential disinfectant resistance as it is assumed that the recommended concentration of disinfectants is always well above the level required to kill the target organism.”
Biofilm can form in the long flexible channels of endoscopes, but also in cracks or on broken components of the instrument. Vigorous adherence to all reprocessing steps—especially precleaning at bedside—is needed to help limit the risk of cross-contamination. Surveillance testing is recommended by international groups as another way to ensure successful reprocessing.
The disposable components now being recommended by the FDA for duodenoscopes are another step towards mitigating the risk of infection transmission during endoscopy.
Single-use bronchoscopes, however, are available and can greatly reduce any risk of cross-contamination.
While rare, cases of cross-contamination during bronchoscopy do happen. It’s also likely that many cases of device-related infection go unreported due to poor surveillance. Because of that, it’s vital that reprocessing staff be aware of MDRO infection risks and the ways to prevent biofilm from forming on flexible endoscopes.