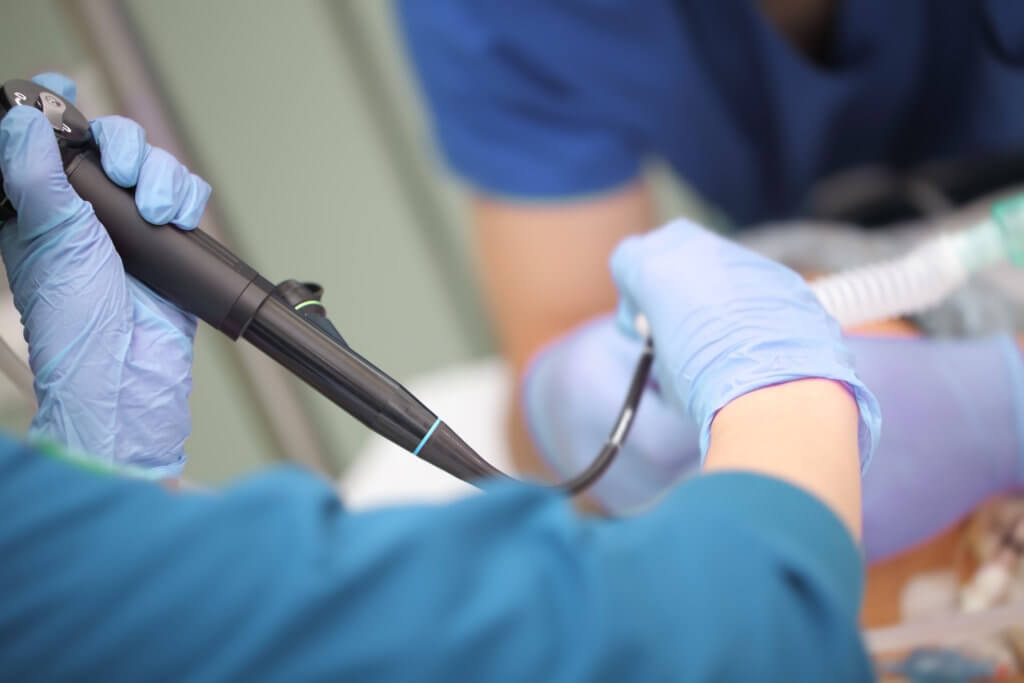
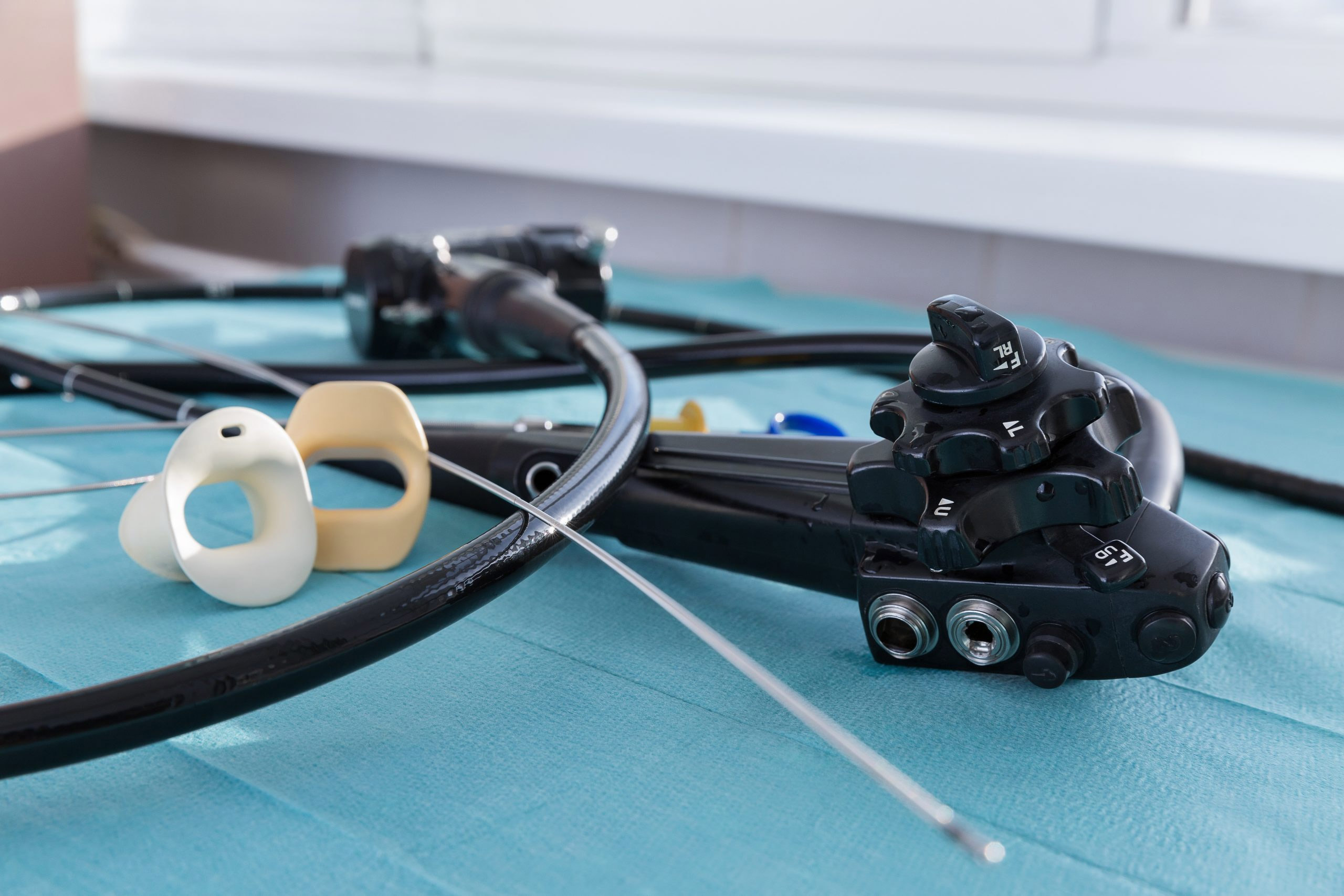
For the first time in almost a decade, the ECRI Institute, in its annual ranking of the top health technology hazards, shifted its focus from the threat posed by improperly cleaning endoscopes to the risk of recontamination due to the improper handling of an endoscope after it has been cleaned.
Mishandling flexible endoscopes after disinfection, leading to patient infections, came in at No. 5 on the list.
Flexible endoscopes are notoriously difficult to clean and disinfect, given their long narrow channels and delicate nature. Manufacturers offer varying guidelines for cleaning, disinfecting and reprocessing scopes, and industry guidelines for reprocessing endoscopes include more than 100 steps. The process can take more than two hours to complete.
But, as the ECRI Institute recognized in its most recent hazards list, the threat of recontamination remains even after endoscopes are cleaned and disinfected. Endoscopes must be handled and stored properly after reprocessing and before reuse to avoid recontamination.
Bronchoscopes are one of the most commonly-used instruments in a hospital, enabling doctors to see the airways to the lungs and perform interventions such as lung biopsies or removing pus and tumors. Reusable bronchoscopes also pose a high risk of cross-contamination.
As robust as industry guidelines are when it comes to cleaning, disinfecting and reprocessing endoscopes, suggested protocols for drying and storing endoscopes are noticeably lacking. The long flexible channels of endoscopes, if not dried properly, can foster microbial growth, even if only stored for a short time. In fact, some health care-associated infections of P. aeruginosa, S. marcescens, M. tuberculosis, and M. chelonae have been traced back to improper ethanol flushings or not following a correct drying procedure.
ECRI’s 2019 Health Technology Hazards report offers some guidelines for preventing the recontamination of flexible endoscopes after reprocessing and disinfection, including “purging endoscope channels with clean air at the end of the reprocessing process” and transporting endoscopes in a “clean enclosed container, dedicated to that purpose.”
Personal protective equipment is also mentioned as necessary for ensuring endoscopes aren’t compromised, especially between reprocessing and high-level disinfection.
Each year, the institute compiles its health tech hazards list by identifying “the potential sources of danger that we believe warrant the greatest attention for the coming year.” It decides which technologies deserve the most attention that year—a list that does not necessarily include the most frequently-reported technology-related problems in the healthcare space.
“All the items on our list represent problems that can be avoided or risks that can be minimized through the careful management of technologies,” the ECRI Institute stated on the 2019 report.
Flexible endoscopes have made the list every year since 2010, though in 2019 the focus shifted from improper cleaning of scopes to the mishandling of flexible endoscopes after they have been disinfected. Cross contamination and flexible endoscopes topped their lists in 2010 and 2016. “Less well-known is that improper handling of storage practices can recontaminate previously disinfected scopes, heightening the risk of patient infections,” this year’s report noted.
For more information about industry guidelines regarding endoscope drying and storage, visit www.bedsidebronchoscopy.com/guidelines.

Reference Links: